To overcome the challenges posed by the development of high-stability, high-conductivity electrolytes, we have divided this theme into three scientific opportunities:
A1: Reducing operating temperatures for oxygen ion conducting electrolytes via strain engineering.
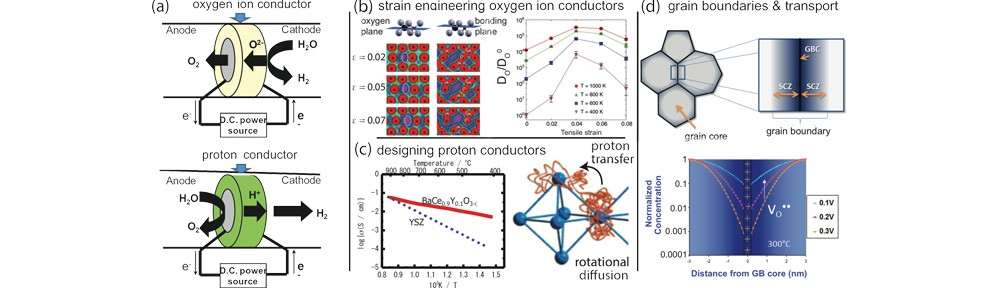
The traditional electrolyte for oxygen ion conducting SOECs is Yttria-stabilized Zirconia (YSZ). Although these systems exhibit the highest efficiency among water electrolysis schemes to date the current limitation is the high operating temperature (700-900°C) required to achieve sufficient ion conductivity (>10-2 S/cm), which leads to rapid degradation and short operating lifetimes.
A2: Establishing design principles for high proton conductivity: What determines whether a given material is an oxide ion conductor or a proton conductor?
We will establish avenues for lowering the operating temperatures of oxygen ion electrolytes. In contrast to oxygen ion electrolytes, the temperature dependence of the conductivity for proton conducting electrolytes is generally lower due to the lower activation energy of proton motion. The current limitation for these systems is, however, that the materials are thermodynamically unstable and often decompose when exposed to steam at operating temperatures around 500°C. Currently, there is no satisfactory proton conducting oxide for steam electrolysis at 500°C.
A3: Tuning grain boundary structure and chemistry for optimal transport of ions: interfaces by design in nanostructured materials.
As SOEC components become increasingly nanostructured (e.g., thin film electrolytes and high surface area nanoporous electrodes), the grain size necessarily decreases and the density of internal interfaces (grain boundaries) dramatically increases. Grain boundaries therefore contribute significantly to the transport properties, and their structure and chemistry need to be optimized.
<Back to Research Page